Dialing in spring nutrition: A plan for annuals
Fertilizer decisions go far beyond just feeding plants. Nitrogen form, phosphorus levels and micronutrient management all play a role in controlling growth and improving crop quality.

Strategic fertility choices to improve growth control and crop quality in the greenhouse
Key Takeaways
- Nitrogen concentration and form are key tools for controlling plant growth
- Ammonium lowers pH; nitrate raises pH in the root zone
- Phosphorus restriction helps reduce plant stretch
- Fertilizer strategies should vary based on crop type and production stage
- Micronutrient needs differ widely across species
- Managing substrate pH is critical for nutrient availability
Plants need 12 different mineral nutrients to complete the life cycle. However, when it comes to production horticulture, some nutrients warrant more attention than others. There are three important considerations in any annual bedding plant fertilization program: nitrogen (form and concentration), phosphorus and micronutrients.
NITROGEN
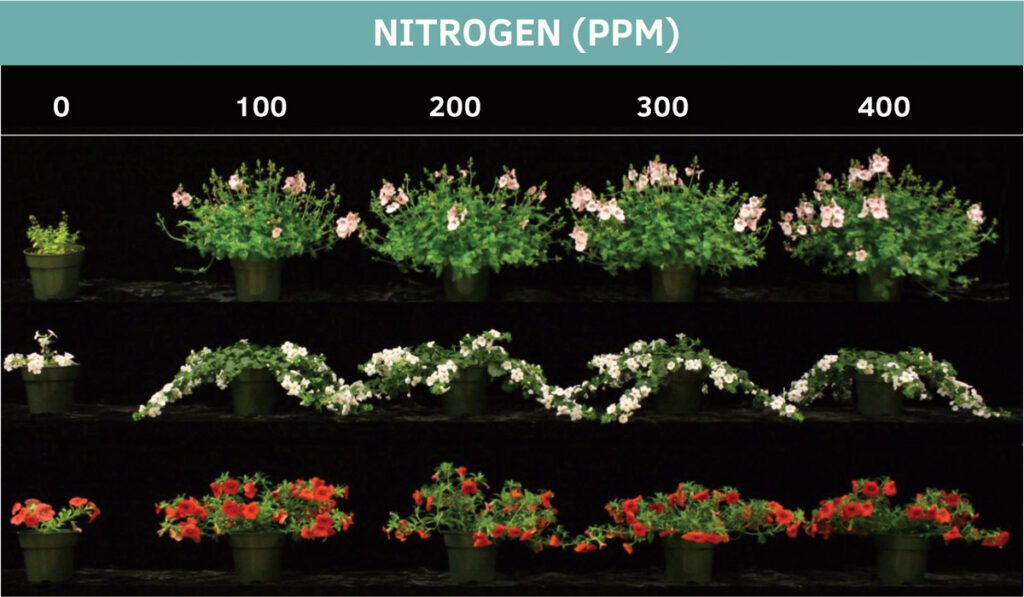
One of the primary considerations with fertilizing spring annuals is the nitrogen (N) concentration applied to crops. Of course, there is variation across crops, from those requiring minimal fertilization such as New Guinea impatiens, to those with greater requirements, like zonal geraniums and petunias. But within these relative nutrient requirements, manipulating N concentrations is an effective way to help control plant growth (Fig. 1).
Increasing N can stimulate growth, sometimes to an unwanted degree, and requires growth control from plant growth retardants. Alternatively, restricting excessive N can curb excessive growth and help tone crops. The ability to change N concentration at each fertilization is one of the biggest advantages to using water- soluble fertilizers with adjustable-ratio fertilizer meters. Being able to respond to crop needs based on changes in the growing environment or other cultural factors gives growers the ability to control their crops.
Form matters
In addition to nitrogen concentrations influencing plant growth, the type or form of nitrogen is important to consider. Nitrogen in fertilizers can be classified as one of two different forms: ammonium (NH4 +) or nitrate (NO3 -) nitrogen. Urea is considered an ammoniacal form of nitrogen since it is converted to NH4 + in substrate.
There are two primary reasons we consider the nitrogen form when selecting fertilizers: nitrifying bacteria and pH effects. First, NH4 + is converted to NO3 – by nitrifying bacteria. However, the optimal temperature for these nitrifying bacteria is 77° F. As temperatures decrease below that, the conversion process is slowed, with activity cut in half by 64° F and minimized by 50° F.
The pH effects of NH4 + and NO3 – are also important considerations when developing fertilizer programs. When NH4 + is taken up by plant roots, a proton or hydrogen ion (H+) is released into the substrate to maintain an appropriate electrochemical balance in the root — positively charged ion in (NH4 +), positively charged ion out (H+). However, the H+ released into the substrate also causes the root zone pH to decrease as it becomes more acidic.
Alternatively, when NO3- is taken up by roots, a hydroxide (OH-) is released — negatively charged ion in (NO3 -), negatively charged ion out (OH-) — and this causes the pH in the root zone to increase as it becomes more basic.
The effect of the N form on substrate pH is reflected by the potential acidity for formulations that can decrease pH, or potential basicity for those formulations that can increase pH.
So what balance of NH4 + to NO3 – should you look for?
First, for cool production temperatures, select a fertilizer with a greater proportion of NO3 -. This helps avoid any damage from ammonium toxicity that could occur when nitrifying bacteria aren’t converting NH4 + to NO3 – quickly enough.
At warmer temperatures, both NH4+- and NO3 –based formulations can be used with no concern.
With respect to effects on pH, select a potentially acidic fertilizer for crops requiring a lower rootzone pH or a potentially basic fertilizer for crops requiring a higher rootzone pH. We will get more in-depth with that later.
PHOSPHORUS
Phosphorus (P) is the second macronutrient that deserves serious consideration when fertilizing containerized annuals, as it can promote stretch and unwanted growth. By restricting the amount of P applied to crops, you can inhibit plant growth to produce more appropriately sized plants (Fig. 2).
While this strategy is already widely accepted and used for producing compact seedling plugs, it is also effective on containerized flowering plants, and the effectiveness of P restriction is catching on.

Strategies for use
There are two strategies for restricting the amount of P provided to crops during finishing. First, select fertilizers with low phosphorus relative to nitrogen. When we look at a per-ppm N basis, less P is provided to plants when using 15-5-15 or 13-2-13 fertilizers compared to 20-10-20 fertilizer. This approach requires several different fertilizer formulations to vary the amount of P relative to N.
Another approach is to decouple N and P altogether and select a fertilizer without P, adding it in as needed at any concentration from monopotassium phosphate (MKP), independent of the amount of N provided.
Materials matter
Also consider the material that you are starting with — seedling plugs or rooted cutting liners — since this can influence your P restriction strategy. There is much more stored P in larger vegetative liners compared to smaller seedling plugs. As a result, more P can be withheld from the rooted cuttings during finishing than the seedling plugs following transplanting. If the idea of restricting phosphorus raises concerns about flowering, it shouldn’t. The amount of P required for healthy and numerous flowers is relatively small, especially for crops with short production times like most spring annuals produced commercially.
MICRONUTRIENTS
The final consideration for fertilizing annual bedding plants is the micronutrient requirements.
We see a wide variety in requirements across greenhouse crops, and spring annuals are no different. Some crops don’t have any special micronutrient requirements and should be grown at a pH of 5.8 to 6.2.

The effects of pH
Crops like petunia and calibrachoa require more micronutrients and should be grown at a lower pH from 5.4 to 5.8 to maximize micronutrient availability and/or provided with higher concentrations of micronutrients in the fertilizer (Fig. 3).
Alternatively, crops like geranium and marigold require less micronutrients and should be grown at a higher pH from 6.2 to 6.6 to restrict micronutrient availability and/or provided with lower concentrations of micronutrient in the fertilizer.
Avoiding micronutrient disorders, deficiencies or toxicities are best managed by keeping rootzone pH in check. As discussed earlier, using a potentially acidic fertilizer is one approach to keeping substrate pH low for micronutrient-inefficient species, though reducing limestone in substrate or increasing water acidification may be needed.
Alternatively, micronutrient-efficient species should be fertilized with basic fertilizers to increase pH, while increasing substrate lime or decreasing water acidification can help maintain an elevated pH.
Fertilizer concentrations
With respect to managing micronutrient concentrations in fertilizers, an approach like that outlined for P is taken. First, you can manage the concentration of micronutrients applied to crops by selecting fertilizers with different ratios of micronutrients relative to N.
On a per-ppm N basis, Peat Lite formulations of 20-10-20 and 15-5-15 have twice the micronutrient concentration compared to the original or general- purpose formulations of the same analyses. Some fertilizers, such as Jacks 18-2-18, have twice the micronutrient concentration of the Peat Lite formulations.
For micronutrient-efficient species, select fertilizers with lower micronutrient concentration formulations, and higher micronutrient concentration formulations. Just like P, it can be useful to separate N (and other macronutrients) from the micronutrients. By using a macronutrient-only fertilizer, combined with a micronutrient-only mix like STEM (soluble trace element mixture) or MOST (mix of soluble traces), any micronutrient concentration can be achieved relative to any N concentration — the ultimate flexibility.
Fertilizer programs for annuals aren’t simple. But focusing on three key aspects of mineral nutrition — nitrogen, phosphorus, and micronutrients — will minimize challenges and maximize benefits. Simplifying success in the spring is something we can all enjoy. Learn how nitrogen, phosphorus and micronutrient strategies impact greenhouse crop growth, pH control and overall plant quality.
FAQ
Why is nitrogen important for growth control?
Nitrogen levels directly influence plant growth rate and can be adjusted to manage size and vigor.
What is the difference between ammonium and nitrate nitrogen?
Ammonium lowers substrate pH, while nitrate raises it.
Does phosphorus affect plant height?
Yes, higher phosphorus can promote stretch, while restricting it helps produce compact plants.
Why is pH important in fertilization?
pH affects nutrient availability and can lead to deficiencies or toxicities if not managed properly.
Can micronutrients be adjusted independently?
Yes, growers can use separate micronutrient mixes to fine-tune concentrations.
Christopher Currey is an associate professor in the department of horticulture at Iowa State University. He can be reached at ccurrey@iastate.edu.