
From lab to leaf: Measuring biocontrol quality
Editor’s note: Below is the condensed article as printed in the July 2025 edition of Greenhouse Product News magazine. Please find the full version after the images.
Determining the number of live individuals (quality assessment) of commercially available biological control agents (BCAs) — including parasitoids, predators and entomopathogenic nematodes — is important to ensure the success of a biological control program.
After releasing BCAs into greenhouses, growers should determine if they are viable by sampling the products, such as slow release sachets, release cards or containers). Inferior quality may lead to the false assumption that biological control is not effective in managing pest populations because pest counts remained at plant damaging levels, even after releasing BCAs.
Furthermore, the quality of BCAs purchased from distributors/suppliers should be assessed after releasing into production systems to determine if enough live individuals are present to maintain pest populations below plant damaging levels. The quality of the same BCA can differ depending on the commercial distributor/supplier.
The success of a biological control program depends on the quality of the BCAs released. Proper package handling of BCAs — including protection from extreme temperatures and relative humidity — is crucial for keeping products alive.
COMMON PRODUCTS
Neoseiulus cucumeris is a predatory mite of the western flower thrips. It is commercially available in slow-release sachets (SRS) that contain eggs, larvae, nymphs and adults mixed with a bran carrier. SRS are placed among greenhouse-grown crops. Adults will emerge from perforated holes and disperse among crops over 4 to 6 weeks.
Encarsia formosa and Eretmocerus eremicus are parasitoids of the greenhouse whitefly (Trialeurodes vaporariorum) and sweetpotato whitefly (Bemisia tabaci). They are commercially available as release cards (RC), each with a predetermined number of parasitized whitefly pupae.
Limited information is available on the quality of BCAs delivered by distributors/suppliers and released into greenhouse production systems. This study aimed to assess the quality of shipments of the predatory mite, N. cucumeris, and the whitefly parasitoids, E. formosa and E. eremicus, received from a distributor/supplier throughout the 2023 growing season in a greenhouse production system.
EXPERIMENTAL APPROACH
The study was conducted during the 2023 growing season at 5-H Greenhouses in Wamego, Kansas, and involved the production of bedding plants (e.g., ornamentals and vegetables), chrysanthemum (Tanacetum x grandiflorum) and poinsettia (Euphorbia pulcherrima). Eight shipments of BCAs were received, and all were ordered and obtained from a distributor/ supplier in the U.S.
Upon receipt, five SRS and five RC were randomly selected from each box and placed into 15 x 15 x 5-mm sealed plastic containers and stored in a refrigerator set at 37.9° F until they were collected within 24 hours. The samples were then transported to a laboratory in the Department of Entomology at Kansas State University in Manhattan, Kansas. Laboratory conditions were maintained at 66.2° F to 71.6° F and 50% to 60% relative humidity under constant light. The SRS containing N. cucumeris were used during bedding plant crop production. The RC containing E. formosa were used for a tomato (Solanum lycopersicum) crop, while the RC containing E. eremicus were used during poinsettia crop production.
The study was set up as a completely randomized design.
QUALITY ASSESSMENT
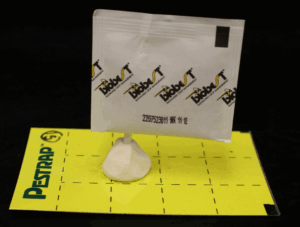
Five SRS with Neoseiulus cucumeris were randomly selected from each box of 1,000 to determine the quality of each shipment. The remaining SRS were placed among the bedding plant crops. A single SRS was inserted into a mold of clay placed on a 2.9 x 5-inch yellow sticky card (Figure 1). The adults that emerged from the SRS and captured on the yellow sticky card were counted using a dissecting microscope at 5 to 27 days following initial preparation of the quality assessment.
Five RC with Encarsia formosa and Eretmocerus eremicus were randomly selected from each box of 100 to determine the quality of each shipment. The remaining RC were placed among the tomato and poinsettia crops. A single RC — containing 50 E. formosa or 100 E. eremicus parasitized whitefly pupae — was placed into a 7.6-ounce clear, wide-mouth Mason jar (Figure 2). A 1 x 1-inch yellow sticky square was attached to the underside of the lid using double-sided sticky foam. The adults captured on the yellow sticky square were counted using a dissecting microscope at 7, 13 and 17 days following initial preparation of the quality assessment. The number of E. eremicus adults were counted at 8 to 24 days following initial preparation of the quality assessment.
WHAT IT ALL MEANS
Quality assessment helps confirm that commercially available BCAs are alive when released into greenhouse production systems. Greenhouse producers may not know how many individuals are expected to emerge from SRS or RC provided by the distributor/supplier. Consequently, if too few emerge, the BCAs will not manage insect pests below plant-damaging levels. Distributors/suppliers are responsible for providing live BCAs, but they have no control over the shipping process, which can impact product quality. Greenhouse producers should conduct quality assessments after releasing BCAs, using the procedures described in our study for SRS and RC.
In our study, emergence rates from SRS and RC varied throughout the growing season depending on the shipment date.
Product information indicates that predatory mites should begin emerging from the SRS within 3 to 4 weeks, with full emergence after 6 weeks. Each SRS is supposed to contain 1,000 predatory mites. However, the number of N. cucumeris adults that emerged from the SRS during the assessment period (days) and captured on the yellow sticky cards was well below 1,000 by the final assessment date.
High-quality BCAs are key to the success of biological control programs. If a quality assessment indicates that not enough live individuals are emerging from the SRS or RC, then the distributor/supplier should be contacted immediately. In the meantime, greenhouse producers may need to apply an insecticide to manage pest populations, which negates implementing a biological control program.
Neoseiulus cucumeris adults that emerged from the SRS and captured on the yellow sticky card were counted using a dissecting microscope for the quality assessment. Photos courtesy of Raymond Cloyd.
The number of E. eremicus adults were counted at 8 to 24 days following initial preparation of the quality assessment
The full version of this study is available below.
Quality assessment of biological control agents in a greenhouse production system
Quality assessment (determining the number of live individuals) of commercially available biological control agents (BCAs), including parasitoids, predators and entomopathogenic nematodes, is important to ensure the success of a biological control program. After releasing BCAs into greenhouses, growers should determine if the individuals received from distributors/suppliers are viable by sampling the products (e.g., slow-release sachets (SRS), release cards (RC) or containers). BCAs of inferior quality may lead to a false presumption that biological control is not effective in managing insect and/or mite pest populations when pest populations remain at plant damaging levels, even after releasing biological control agents. Therefore, the success of a biological control program depends on the quality of the BCAs released. Furthermore, to ensure continued business from customers, distributors/suppliers must properly package BCAs to provide protection from exposure to extremes in temperature and relative humidity encountered during shipping.
The quality of BCAs is not always associated with the distributor/supplier but instead may be related to shipping, which is beyond their control. For example, exposure to unfavorable environmental conditions, such as temperature and relative humidity, can affect the number of live BCAs received by a greenhouse producer, and the quality can vary depending on the packaging of products from distributors/suppliers. In addition, the quality of the same BCAs can differ depending on the commercial distributor/supplier. Consequently, the quality should be assessed after releasing them into greenhouse production systems to determine if enough live individuals are present to maintain pest populations below plant damaging levels.
Neoseiulus cucumeris is a predatory mite of the Western flower thrips, Frankliniella occidentalis. N. cucumeris is commercially available in slow-release sachets that contain eggs, larvae, nymphs and adults mixed with a bran carrier. Slow-release sachets are placed among greenhouse grown crops. Adults emerge from the sachets through perforated holes and then disperse among crops within the greenhouse over 4 to 6 weeks.
Encarsia formosa and Eretmocerus eremicus are parasitoids of the greenhouse whitefly, Trialeurodes vaporariorum and sweetpotato whitefly, Bemisia tabaci. The parasitoids are commercially available as release cards with a predetermined number of parasitized whitefly pupae attached to each release card.
There is minimal information on the quality of BCAs received from distributors/suppliers that are released into greenhouse production systems. Therefore, the objective of our study was to assess the quality of shipments of the predatory mite, N. cucumeris, and the whitefly parasitoids, E. formosa and E. eremicus, received from a distributor/supplier throughout the growing season in a greenhouse production system in 2023. Our study would help greenhouse producers determine the number of viable individuals released into greenhouses.
Materials and methods
This study was conducted during the growing season of 2023 that involved the production of bedding plants (e.g., ornamentals and vegetables), chrysanthemum (Tanacetum x grandiflorum), and poinsettia (Euphorbia pulcherrima) at 5-H Greenhouses in Wamego, Kansas. There were eight shipments of BCAs received, and all were ordered and obtained from a distributor/supplier in the U.S. The slow-release sachets (SRS) were shipped in a box with brown paper and the release cards (RC) were shipped in a Styrofoam container with ice packs and brown paper. Upon receipt by the greenhouse producer, 5 SRS and 5 RC were randomly selected from each box and placed into 15 x 15 x 5 millimeter [0.59 x 0.59 x 0.19 inch (length x width x height)] sealed plastic containers. The sealed plastic containers were stored in a refrigerator set at 37.9° F (3.3° C) until they were collected within approximately 24 hours. After collecting the SRS and/or RC, they were transported to a laboratory in the Department of Entomology at Kansas State University in Manhattan, Kansas. The laboratory conditions were 66.2° F to 71.6° F (19.8° C to 22° C) and 50% to 60% relative humidity (RH) under constant light. The SRS containing N. cucumeris were used during production of the bedding plant crops. The RC containing E. formosa were used for a tomato, Solanum lycopersicum, crop and the RC containing E. eremicus were used during production of the poinsettia crop.
The study was set up as a completely randomized design. Boxes containing 1,000 SRS of the predatory mite, N. cucumeris, were received on March 3 and 26, April 26, May 11, and July 20, 2023. Boxes containing 100 RC of E. formosa were received on Jan. 18, 2023, and 100 RC of E. eremicus were received on Sept. 15 and Oct. 25, 2023. Each box of RC associated with E. formosa indicated 50 parasitized greenhouse whitefly pupae per RC and each box of RC affiliated with E. eremicus indicated 100 parasitized sweetpotato whitefly pupae per RC.
Quality assessment of Neoseiulus cucumeris
Five SRS were randomly selected from each box of 1,000 to determine the quality of each shipment. The remaining SRS were placed among the bedding plant crops. A single SRS was inserted into a mold of clay placed on a 7.6 x 12.7 centimeter (2.9 x 5.0 inch) yellow sticky card (Figure 1). The number of adults that emerged from the SRS and were captured on the yellow sticky card were counted using a dissecting microscope. The number of adults were counted on the yellow sticky card 5 to 27 days following initial preparation of the quality assessment.
Quality assessment of Encarsia formosa and Eretmocerus eremicus
Five RC were randomly selected from each box of 100 to determine the quality of each shipment. The remaining RC were placed among the tomato and poinsettia crops. A single RC, containing 50 E. formosa or 100 E. eremicus parasitized whitefly pupae per RC, was placed into a 225 milliliter (7.6 fluid ounces) clear glass wide mouth Mason jar (Figure 2). A 2.5 x 2.5 centimeter (1.0 x 1.0 inch) yellow sticky square was attached to the underside of the lid using double-sided sticky foam. The number of adults captured on the yellow sticky square were counted using a dissecting microscope. The number of E. formosa adults were counted on the yellow sticky square after 7, 13 and 17 days following initial preparation of the quality assessment. The number of E. eremicus adults were counted on the yellow sticky square 8 to 24 days following initial preparation of the quality assessment.
Data collection and analysis
There were eight shipments of biological control agents. Costs associated with each of the BCA shipments were determined based on invoices from the distributor/supplier. Data related to the number of adults that emerged from the SRS and RC, and were captured on the yellow sticky card or square, were summarized using descriptive statistics. Data from shipments 1 (SRS of N. cucumeris), 4 (SRS of N. cucumeris), 6 (RC of E. formosa) and 7 (RC of E. eremicus) were analyzed using a Student’s t-test. Data from shipments 2 (SRS of N. cucumeris), 5 (SRS of N. cucumeris) and 8 (RC of E. eremicus) were analyzed using an analysis of variance. Data related to shipments 3 and 4 were not analyzed because only 1 box was received.
Results
Results from the SRS containing N. cucumeris are presented in Tables 1 to 5, and results from the RC containing E. formosa or E. eremicus are presented in Tables 6 to 8.
Quality assessment of Neoseiulus cucumeris
There were significant differences in the number of N. cucumeris adults that emerged from the SRS associated with the different boxes for shipments 2 (F=39.29; df=2, 23; P<0.001) and 4 [t(8)=-6.26; P=0.0002), but not for shipments 1 [t(28)=-0.18; P=0.85] and 5 (F=2.46; df=2, 38; P=0.098). For shipment 2, significantly more N. cucumeris adults emerged from the SRS affiliated with the second box [250.0 ± 10.8; mean ± standard error of the mean (SEM)] than the first (147.1 ± 9.2) and third (214.7 ± 2.9) boxes.
The number of N. cucumeris adults that emerged from the SRS varied depending on the time of year during the growing season. The mean and SEM of adults that emerged from the SRS on the final assessment date for all 5 shipments was between 156.2 ± 12.9 and 410.0 ± 24.2, and the range was between 132 and 469 (Tables 1 to 5).
Quality assessment of Encarsia formosa and Eretmocerus eremicus
There was no significant difference [t(28)=0.71; P=0.48] in the number of E. formosa adults that emerged from the RC affiliated with the different boxes for shipment 6. There were significant differences in the number of E. eremicus that emerged from the RC from the different boxes for shipment 7 [t(28)=-2.38; P=0.024] and 8 (F=4.02; df=2, 30; P=0.028). For shipment 7, significantly more E. eremicus adults emerged from the RC affiliated with box 2 (68.3 ± 5.4) than box 1 (53.7 ± 2.9). For shipment 8, significantly more E. eremicus adults emerged from RC from box 1 (47.0 ± 3.4) than box 2 (33.6 ± 3.2), but there were no significant differences in the number of E. eremicus adults that emerged from RC from box 1 (47.0 ± 3.4) and 3 (41.2 ± 3.7), and box 2 (33.6 ± 3.2) and 3 (41.2 ± 3.7).
The mean, SEM, and range of E. formosa adults that emerged from the RC associated with the one shipment of 2 boxes was between 52.6 ± 1.2 (range: 49 to 55) and 47.8 ± 2.2 (range: 42 to 53) (Table 6). The mean (± SEM) of E. eremicus adults that emerged from the RC from the two shipments of 5 boxes was below the 100 indicated on the box from the distributor/supplier (40.0 ± 5.0 and 75.2 ± 9.4), with the range between 24 and 94 (Tables 7 and 8).
The quantity, biological control agent, unit cost, total cost of BCA products and freight charge of the individual shipments (boxes) from the distributor/supplier are presented in Table 9.
Discussion
Quality assessment is important to confirm that commercially available BCAs, such as parasitoids and predators, are alive when released into greenhouse production systems. Our study demonstrates that the quality, based on the number of live biological control agents from a distributor/supplier, can vary during the growing season. Greenhouse producers may not know the number of individuals that should emerge from SRS or RC provided by the distributor/supplier. Consequently, if not enough individuals emerge, then the BCAs will not manage insect pests below plant damaging levels.
Distributors/suppliers are responsible for providing live individuals of BCAs. However, distributors/suppliers have no control of the shipping process, which can affect the quality of BCA shipments. Therefore, greenhouse producers should conduct quality assessments after releasing BCAs by following the procedures described in our study for SRS and RC.
Studies indicate there are issues associated with BCA shipments received from distributors/suppliers, such as the number of individuals that emerge and survival of BCAs. In our study, the number of individuals that emerged from the SRS and RC varied during the growing season depending on the shipment date.
Information indicates that the predatory mites should have emerged from the SRS after 3 to 4 weeks with all the predatory mites having emerged from the SRS after 6 weeks. It is important that enough N. cucumeris adults emerged from the SRS to manage Western flower thrips populations below plant damaging levels because N. cucumeris only feeds on the first instar larvae. Hence, if there is any delay in emergence, or if not enough N. cucumeris adults emerged from the SRS, then management of Western flower thrips populations will be compromised. There are supposed to be 1,000 predatory mites in each SRS. However, the number of N. cucumeris adults that emerged from the SRS during the assessment period (days) and were captured on the yellow sticky cards was well below 1,000 with means (± SEM) between 156.2 ± 12.9 and 410.0 ± 24.2 for all 5 shipments on the final assessment date. The invoices from the company indicated that there were “250/sachet.” However, what does the “250/sachet” mean? Is this the number of predatory mites per SRS? Is this the number of adults? What about the number of eggs, larvae, and nymphs? There were several instances in which over 250 adult predatory mites emerged from the SRS (Tables 1 through 5). Consequently, distributors/suppliers need to provide information to greenhouse producers on the number of predatory mites that should emerge from each SRS.
The SRS contain eggs, immatures (larvae and nymphs) and adults of N. cucumeris mixed with a bran carrier. In addition, the SRS contain bran mites, Tyrophagus putrescentiae, as a temporary food source. The bran mites emerged from the SRS and were captured on the yellow sticky card, thus increasing the difficulty in accurately counting the number of N. cucumeris adults. Approximately 30% of the mites that were captured on the yellow sticky card were bran mites, which may have resulted in an overestimation of the number of N. cucumeris counted.
The mean (± SEM) of E. formosa adults that emerged from the RC on the final assessment date was 52.6 ± 1.2 and 47.8 ± 2.2, which was near the 50 indicated on the box from the distributor/supplier. However, the mean (± SEM) of E. eremicus adults that emerged from the RC on the final assessment date was below the 100 (40.0 ± 5.0 to 75.2 ± 9.4) indicated on the box from the distributor/supplier. Consequently, additional RC were purchased and placed among the poinsettia crop to avoid having to manage high numbers of whiteflies, which would have affected marketability and salability.
We collected five samples of SRS and RC from each shipment (box or boxes). However, five samples may not have been enough to accurately estimate the quality of the BCA shipments. Nonetheless, significant differences were found despite the small sample size. Moreover, our study attempted to simulate what would be practical for a greenhouse producer to conduct quality assessments of BCAs.
A major expense when purchasing BCAs is the freight charge because products must be shipped overnight to ensure survival upon receipt. Nonetheless, the freight charge can sometimes be higher than the actual cost of the BCAs. Freight charges can be a deterrent in implementing a biological control program, especially when shipments are scheduled to arrive weekly. In our study, the total freight charge for all eight shipments of the BCAs was $1,013.23, which is $543.27 less than the total cost ($1,556.50) of the BCAs.
In conclusion, our study demonstrates that greenhouse producers should conduct quality assessments to determine that BCAs purchased from a distributor/supplier are viable when released among greenhouse grown horticultural crops. The quality (number of live individuals) of BCAs is important for the success of biological control programs. If a quality assessment indicates that not enough live individuals are emerging from the SRS or RC, then the distributor/supplier should be contacted immediately to receive a refund or a new shipment. In the meantime, the greenhouse producer may have to apply an insecticide to manage or maintain an insect pest population below plant damaging levels, which negates implementing a biological control program.
Table 1. Recorded count data, mean and standard error of the mean (SEM) of Neoseiulus cucumeris adults that emerged from the slow-release sachets (SRS) and were captured on a 7.6 x 12.7 centimeter (2.9 x 5 inch) yellow sticky card on March 11, 21 and 27, 2023. Slow-release sachets were prepared for quality assessment on March 7, 2023. There were 2 boxes of 1,000 with five SRS sampled from each box.
| Box 1 | March 11, 2023
(5 days)* |
March 21, 2023
(15 days) |
March 27, 2023
(21 days) |
| SRS Number | |||
| 1 | 33 | 288 | 320 |
| 2 | 90 | 246 | 367 |
| 3 | 82 | 268 | 286 |
| 4 | 84 | 263 | 294 |
| 5 | 65 | 225 | 284 |
| Mean ± SEM | 70.8 ± 10.3 | 258.0 ± 10.6 | 310.2 ± 15.5 |
| Box 2 | |||
| SRS Number | |||
| 1 | 49 | 250 | 316 |
| 2 | 46 | 390 | 392 |
| 3 | 51 | 305 | 350 |
| 4 | 45 | 285 | 297 |
| 5 | 30 | 250 | 264 |
| Mean ± SEM | 44.2 ± 3.7 | 296.0 ± 25.7 | 323.8 ± 22.0 |
* Number of days that predatory mites were counted on yellow sticky cards following initial preparation (March 7, 2023).
Table 2. Recorded count data, mean and standard error of the mean (SEM) of Neoseiulus cucumeris adults that emerged from slow-release sachets (SRS) and were captured on a 7.6 x 12.7 centimeter (2.9 x 5 inch) yellow sticky card on April 10 and 21, 2023. Slow-release sachets were prepared for quality assessment on March 27, 2023. There were 3 boxes of 1,000 with 5 SRS sampled from each box.
| Box 1 | April 10, 2023
(15 days)* |
April 21, 2023
(26 days) |
| SRS Number | ||
| 1 | 121 | 132 |
| 2 | 137 | 142 |
| 3 | 105 | 132 |
| 4 | 144 | 185 |
| 5 | 183 | 190 |
| Mean ± SEM | 138.0 ± 13.1 | 156.2 ± 12.9 |
| Box 2 | ||
| SRS Number | ||
| 1 | 204 | 290 |
| 2 | 245 | 298 |
| 3 | 212 | 291 |
| 4 | 223 | 234 |
| 5 | 240 | 263 |
| Mean ± SEM | 224.8 ± 7.8 | 275.2 ± 11.9 |
| Box 3 | ||
| SRS Number | ||
| 1 | 206 | 213 |
| 2 | 205 | 207 |
| 3 | 207 | 212 |
| 4 | 224 | 230 |
| 5 | 215 | 228 |
| Mean ± SEM | 211.4 ± 3.6 | 218.0 ± 4.6 |
* Number of days that predatory mites were counted on yellow sticky cards following initial preparation (March 27, 2023).
Table 3. Recorded count data, mean and standard error of the mean (SEM) of Neoseiulus cucumeris adults that emerged from slow release sachets (SRS) and were captured on a 7.6 x 12.7 centimeter (2.9 x 5 inch) yellow sticky card on May 3, 8, and 26, 2023. Slow-release sachets were prepared for quality assessment on April 28, 2023. There was 1 box of 1,000 with 5 SRS sampled from the box.
| Box | May 3, 2023
(6 days)* |
May 8, 2023
(11 days) |
May 26, 2023
(19 days) |
| SRS Number | |||
| 1 | 210 | 250 | 313 |
| 2 | 250 | 252 | 293 |
| 3 | 285 | 310 | 319 |
| 4 | 211 | 230 | 299 |
| 5 | 237 | 264 | 312 |
| Mean ± SEM | 238.6 ± 13.9 | 261.2 ± 13.3 | 307.2 ± 4.8 |
* Number of days that predatory mites were counted on yellow sticky cards following initial preparation (April 28, 2023).
Table 4. Recorded count data, mean and standard error of the mean (SEM) of Neoseiulus cucumeris adults that emerged from slow-release sachets (SRS) and were captured on a 7.6 x 12.7 centimeter (2.9 x 5 inch) yellow sticky card on May 16 and June 6, 2023. Slow-release sachets were prepared for quality assessment on May 11, 2023. There was 1 box of 1,000 with 5 SRS sampled from the box.
| Box | May 16, 2023
(6 days)* |
June 6, 2023
(27 days) |
| SRS Number | ||
| 1 | 222 | 451 |
| 2 | 212 | 330 |
| 3 | 232 | 396 |
| 4 | 288 | 469 |
| 5 | 222 | 404 |
| Mean ± SEM | 235.2 ± 13.5 | 410.0 ± 24.2 |
* Number of days that predatory mites were counted on yellow sticky cards following initial preparation (May 11, 2023).
Table 5. Recorded count data, mean and standard error of the mean (SEM) of Neoseiulus cucumeris adults that emerged from slow-release sachets (SRS) and were captured on a 7.6 x 12.7 centimeter (2.9 x 5 inch) yellow sticky card on July 28, and August 4 and 11, 2023. Slow-release sachets were prepared for quality assessment on July 21, 2023. There were 3 boxes of 1,000 with 5 SRS sampled from each box.
| Box 1 | July 28, 2023
(8 days)* |
August 4, 2023
(15 days) |
August 11, 2023
(22 days) |
| SRS Number | |||
| 1 | 155 | 233 | 354 |
| 2 | 149 | 212 | 333 |
| 3 | 146 | 201 | 310 |
| 4 | 170 | 262 | 363 |
| 5 | 157 | 232 | 305 |
| Mean ± SEM | 124.4 ± 4.1 | 228.0 ± 10.4 | 333.0 ± 11.5 |
| Box 2 | |||
| SRS Number | |||
| 1 | 240 | 331 | 411 |
| 2 | 201 | 254 | 383 |
| 3 | 164 | 197 | 331 |
| 4 | 215 | 251 | 403 |
| 5 | 173 | 204 | 359 |
| Mean ± SEM | 198.6 ± 13.8 | 247.4 ± 23.9 | 377.4 ± 14.6 |
| Box 3 | |||
| SRS Number | |||
| 1 | 219 | 255 | 410 |
| 2 | 283 | 284 | 428 |
| 3 | 192 | 213 | 317 |
| 4 | 227 | 344 | 426 |
| 5 | 268 | 303 | 419 |
| Mean ± SEM | 237.8 ± 16.6 | 279.8 ± 22.0 | 400.0 ± 21.3 |
* Number of days that predatory mites were counted on yellow sticky cards following initial preparation (July 21, 2023).
Table 6. Recorded count data, mean and standard error of the mean (SEM) of Encarsia formosa adults that emerged from release cards (RC) and were captured on a 2.5 x 2.5 centimeter (1 x 1 inch) yellow sticky square on Jan. 27, Feb. 2, and 6, 2023. Release cards were prepared for quality assessment on January 21, 2023. There were 2 boxes of 100 RC per box with each RC containing 50 parasitized greenhouse whitefly, Trialeurodes vaporariorum, pupae per RC. Five RC were sampled from each box.
| Box 1 | January 27, 2023
(7 days)* |
February 2, 2023
(13 days) |
February 6, 2023
(17 days) |
| RC Number | |||
| 1 | 16 | 49 | 49 |
| 2 | 21 | 55 | 55 |
| 3 | 28 | 54 | 54 |
| 4 | 20 | 50 | 50 |
| 5 | 17 | 55 | 55 |
| Mean ± SEM | 20.4 ± 2.1 | 52.6 ± 1.2 | 52.6 ± 1.2 |
| Box 2 | |||
| RC Number | |||
| 1 | 22 | 47 | 47 |
| 2 | 17 | 41 | 42 |
| 3 | 26 | 49 | 53 |
| 4 | 16 | 53 | 53 |
| 5 | 17 | 42 | 44 |
| Mean ± SEM | 19.6 ± 1.9 | 46.4 ± 2.2 | 47.8 ± 2.2 |
* Number of days that adults were counted on yellow sticky squares following initial preparation (Jan. 21, 2023).
Table 7. Recorded count data, mean and standard error of the mean (SEM) of Eretmocerus eremicus adults that emerged from release cards (RC) and were captured on a 2.5 x 2.5 centimeter (1 x 1 inch) yellow sticky square on Sept. 25, Oct. 4 and 9, 2023. Release cards were prepared for quality assessment on Sept. 16, 2023. There were 2 boxes of 100 RC per box with each RC containing 100 parasitized sweetpotato whitefly, Bemisia tabaci, pupae per RC. Five RC were sampled from each box.
| Box 1 | September 25, 2023
(10 days)* |
October 4, 2023
(19 days) |
October 9, 2023
(24 days) |
| RC Number | |||
| 1 | 42 | 52 | 52 |
| 2 | 33 | 47 | 47 |
| 3 | 41 | 58 | 60 |
| 4 | 51 | 68 | 72 |
| 5 | 50 | 65 | 68 |
| Mean ± SEM | 43.4 ± 3.2 | 58.0 ± 3.9 | 59.8 ± 4.6 |
| Box 2 | |||
| RC Number | |||
| 1 | 71 | 85 | 94 |
| 2 | 81 | 93 | 93 |
| 3 | 54 | 66 | 70 |
| 4 | 55 | 77 | 77 |
| 5 | 25 | 42 | 42 |
| Mean ± SEM | 57.2 ± 9.5 | 72.6 ± 8.8 | 75.2 ± 9.4 |
* Number of days that adults were counted on yellow sticky squares following initial preparation (Sept. 16, 2023).
Table 8. Recorded count data, mean and standard error of the mean (SEM) of Eretmocerus eremicus adults that emerged from release cards (RC) and were captured on a 2.5 x 2.5 centimeter (1 x 1 inch) yellow sticky square on Nov. 4, 11 and 18, 2023. Release cards were prepared for quality assessment on Oct. 28, 2023. There were 3 boxes of 100 RC per box with each RC containing 100 parasitized sweetpotato whitefly, Bemisia tabaci, pupae per RC. Five RC were sampled from each box.
| Box 1 | November 4, 2023
(8 days)* |
November 11, 2023
(15 days) |
November 18, 2023
(22 days) |
| RC Number | |||
| 1 | 28 | 49 | 49 |
| 2 | 25 | 58 | 58 |
| 3 | 37 | 60 | 60 |
| 4 | 28 | 43 | 44 |
| 5 | 41 | 62 | 63 |
| Mean ± SEM | 31.8 ± 3.0 | 54.4 ± 3.6 | 54.8 ± 3.5 |
| Box 2 | |||
| RC Number | |||
| 1 | 22 | 36 | 39 |
| 2 | 31 | 53 | 55 |
| 3 | 28 | 44 | 44 |
| 4 | 16 | 37 | 38 |
| 5 | 16 | 22 | 24 |
| Mean ± SEM | 22.6 ± 3.0 | 38.4 ± 5.1 | 40.0 ± 5.0 |
| Box 3 | |||
| RC Number | |||
| 1 | 30 | 44 | 48 |
| 2 | 20 | 52 | 53 |
| 3 | 21 | 37 | 39 |
| 4 | 35 | 62 | 63 |
| 5 | 19 | 47 | 48 |
| Mean ± SEM | 25.0 ± 3.1 | 48.4 ± 4.1 | 50.2 ± 3.9 |
* Number of days that adults were counted on yellow sticky squares following initial preparation (Oct. 28, 2023).
Table 9. Quantity, biological control agent, unit cost, total cost of biological control agent products and freight charge associated with the shipments (boxes) of each biological control agent (Neoseiulus cucumeris, Encarsia formosa, and Eretmocerus eremicus) related to the slow release sachets (SRS) and release cards (RC) received from the distributor/supplier.
| Quantity | Biological Control Agent | Unit Cost | Total Cost | Freight Charge |
| 2 Boxes* | SRS (Neoseiulus cucumeris) | $114.76 | $229.52 | $166.92 |
| 3 Boxes | SRS (Neoseiulus cucumeris) | $114.76 | $344.28 | $250.18 |
| 1 Box | SRS (Neoseiulus cucumeris) | $114.76 | $114.76 | $124.49 |
| 1 Box | SRS (Neoseiulus cucumeris) | $114.76 | $114.76 | $72.98 |
| 3 Boxes | SRS (Neoseiulus cucumeris) | $114.76 | $344.28 | $192.42 |
| 1 Box** | RC (Encarsia formosa) | $28.60 | $28.60 | $82.68 |
| 2 Boxes*** | RC (Eretmocerus eremicus) | $76.06 | $152.12 | $61.78 |
| 3 Boxes | RC (Eretmocerus eremicus) | $76.06 | $228.18 | $61.78 |
| $1,556.50 | $1,013.23 |
* Boxes associated with the slow-release sachets (SRS) of Neoseiulus cucumeris contained 1000 SRS per box.
** Boxes associated with the release cards (RC) of Encarsia formosa contained 100 RC per box.
*** Boxes associated with the release cards (RC) of Eretmocerus eremicus contained 100 RC per box.
Photo courtesy of Raymond Cloyd.










 Video Library
Video Library 


















